 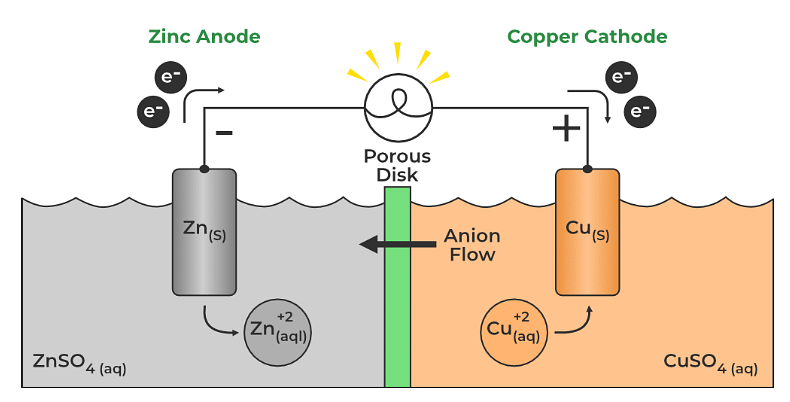 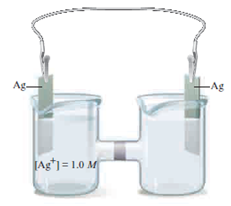
So this is one form of the Nernst equation from the last few videos. Of electrons transferred which is n, times the log of Q. The cell potential, which is what we're trying to find, E, is equal to the standard cell potential E zero, minus. So let's get some more room down here, and let's write down the Nernst equation. How do we find the voltage of our concentration cell? Remember, from the last few videos that the Nernst equation allows us to calculate the potential of the cell. So on the left side, we would have zinc two plus, at initial concentration of 1.0 molar. We can cancel that out, we have two electrons on both sides. So overall, what is happening overall here, so let's draw a line, so we have solid zinc on both sides. So this would be gaining two electrons, to form solid zinc. 
I'll write 1.0 molar concentration, once again, to distinguish More concentrated side, so let's write that, reduction, right here, so this wouldīe zinc two plus ions. It can do that if zinc two plus ions come out of solution. So it needs to decrease the concentration of zinc two plus ions in solution. The more concentrated side needs to decrease its concentration. Those two electrons move in our wire here, and we create a current. 10 molar to distinguish this from the other side, plus two electrons, right, so we lose two electrons, solid zinc loses two electrons to turn into zinc two plus. So we have solid zinc, turning into zinc two plus. Solid zinc turning into zinc two plus is oxidation, so oxidation occurs on the less concentrated side. Into zinc two plus ions, that increases the concentration. So how can we increase the concentration of zinc two plus ions? Well, if solid zinc turned The less concentrated side needs to get more concentrated, so it can do that by increasing the concentration of zinc two plus ions in solution. So how can we make theseĬoncentrations more equal? Let's focus on the lessĬoncentrated side first. There's a tendency for the concentrations to be equalized, and that's enough to create a small voltage. Less concentrated side, and the right side is the The only difference is the concentration. So here we have zincĮlectrode on the left, and zinc electrode on the right. A concentration cell, is a cell that has the sameĮlectrodes on both sides. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
